Vaccinating Dogs Against Lyme Disease: Two Points Of View
Meryl P. Littman
VMD, DACVIM
Dr. Littman is a professor of medicine at the University of Pennsylvania School of Veterinary Medicine. Her research includes canine tick-borne diseases. She received her VMD from University of Pennsylvania.
Richard E. Goldstein
DVM, DACVIM, DECVIM
Dr. Goldstein is chief medical officer at The Animal Medical Center in NYC. His research includes canine Lyme disease and leptospirosis. He received his DVM from Koret School of Veterinary Medicine, Hebrew University of Jerusalem, Israel.
Despite availability of a consensus statement and up-to-date canine vaccination guidelines , the decision whether or not to vaccinate dogs against Lyme disease remains an ongoing debate among many veterinary professionals.
2011 AAHA Canine Vaccination Guidelines.JAAHA.
ACVIM Small Animal Consensus Statement on Lyme Disease in Dogs: Diagnosis, Treatment, and Prevention. Littman MP, Goldstein RE, Labato MA, et al. J Vet Intern Med. 2006 20:422-434. Available at cvm.ncsu.edu/vhc/documents/LymeconsstmtACVIM.pdf.
Todays Veterinary Practice has gathered 3 experts to discuss this topicDr. Meryl Littman and Dr. Richard Goldstein evaluate the benefits and risks of vaccination, while Dr. Richard Ford outlines the current vaccination recommendations.
Theres A Vaccine For Lyme Disease So Why Cant We Get It
Thanks to vaccines, the number of COVID-19 cases has plummeted in the U.S. and restrictions are being lifted across the country. But as we return to our normal activities, we face a more familiar summertime scourge. Were in the thick of Lyme disease season the two-month run from early June to the end of July when 85 percent of infections take place. Surprisingly, vaccines may have allowed us to avoid this epidemic, too. As I learned during my own recent bout with Lyme disease, a vaccine has existed for decades, but its no longer available.
Its yet another frustrating aspect of this mysterious disease. One bite from a tiny, hard-to-detect tick can lead to a host of odd symptoms, including arthritis, serious cardiac issues, and neurological damage in the most severe cases. The disease is easy to treat once you get a diagnosis, but that can be elusive. And while work is underway to develop a new and better vaccine, it may take years to come to market. Heres what we know about how the disease works, and what you can do to stay safe.
Vaccines: Our Greatest Hope In Defeating Aids Ebola Tb And Other Infectious Diseases
Safety concerns began to emerge soon after people began getting LYMErix. Some recipients began to report joint pain and other effects that they attributed to the vaccine. Within a year of the vaccines approval, a class action lawsuit was filed against SmithKline Beecham on behalf of 121 people. Although the suit was eventually settled it provided no compensation to the plaintiffs publicity about it dampened interest in the vaccine.
Meanwhile, growing distrust of vaccines may have compounded LYMErixs struggles.
When a 1998 report in the Lancet identified a purported link between the common MMR vaccine and autism, the study since retracted and considered fraudulent sparked a spike in worry about the safety of vaccines. In October 1999, a new vaccine against rotavirus, the most common cause of diarrhea in infants and young children, was withdrawn from the market after its use was linked to a higher than expected risk of a potentially deadly bowel blockage known as intussusception.
An advisory panel convened in 2001 by the FDA did not find any evidence that LYMErix caused arthritis in humans or an unexpected number of adverse events. But growing suspicion of vaccines in general and publicity about possible side effects had already taken a toll on LYMErixs uptake. SmithKline Beecham projected sales of only 10,000 doses for 2002.
The company withdrew LYMErix in February 2002, citing poor market performance.
Also Check: Vibrant Wellness Lyme Test Reviews
The Lyme Vaccination For Dogs Isnt Foolproof
The shot isnt 100 percent effective, says Dr. Mengel.
But, Dr. Mengel adds, anecdotally, many practices report seeing lower numbers of dogs testing positive for exposure to the bacteria that causes Lyme since using the vaccine in practice over several years.
The Lyme vaccine cannot replace good tick control, because it is not completely effective at preventing Lyme disease, and it does nothing to protect against the many other tick-borne diseases, such as ehrlichiosis and Rocky Mountain spotted fever, says Dr. Brevitz.
Why Not Immunize Everyone
The risk of acquiring Lyme disease is so low in Canada that universal immunization with Lyme disease vaccine is not advisable. In fact, it would not be cost beneficial to immunize everyone even in highly endemic communities in the United States . Therefore, a risk assessment that analyzes the likelihood of exposure to Lyme disease provides the most cost effective method of using this vaccine.
Don’t Miss: Elisabeth Kieselstein Cord Lyme Disease
Constructing A Human Vaccine
Lyme disease is the most prevalent tick-borne disease in North America and Europe. The CDC estimates there are 470,000 clinical diagnosed cases each year in the U.S. alone. Symptoms of Lyme disease vary but can be debilitating, including severe fatigue and headache, neck stiffness, joint pain, heart inflammation and various neurological symptoms.
The cause of Lyme disease is a spiral-shaped bacterium, Borreliella burgdorferi. B.burgdorferi is a highly variable organism, so the key challenge in developing a vaccine is that it has to be able to trigger immune responses in vaccinated individuals that can protect against the diverse strains of the Lyme spirochetes that exist in nature, Marconi says.
Many vaccines work by introducing individual proteins from bacteria, called antigens, to stimulate an antibody response in the body to fight the infection. The canine vaccine, Vanguard crLyme, consists of two proteins: an outer surface protein A , and a custom-designed version of outer surface protein C . OspA was once used in a human vaccine against Lyme disease, called LYMErix. The drug remained on the market for just a few years before being voluntarily discontinued by the manufacturer due to perceived concerns about adverse events.
In light of the stigma associated with the use of OspA in a human vaccine, Marconi made the decision not to use it in a potential vaccine for humans. Im happy to say weve been able to find an improved substitute for OspA, he says.
Discovery And Basic Science
Nonprotein targets are also being assessed for potential utility in Lyme disease vaccine development. The Borrelia do not produce lipopolysaccharide, but do produce glycolipids on their outer surface that may be unique to the Borrelia. During infection, a strong immune response is mounted against some of these nonprotein antigens . One glycolipid in particular, acylated cholesteryl galactoside , has been the focus of investigation as a vaccinogen because of its immunogenicity during infection . ACG induces an IgM antibody response when administered to mice or rabbits in micellar form in PBS, or in DMSO, squalene, or Freunds adjuvants . A protein conjugate of ACG has been synthesized, and is currently being tested for use as a Lyme disease vaccine .
L.H. Sigal, in, 2014
Read Also: Lyme Disease And Liver Damage
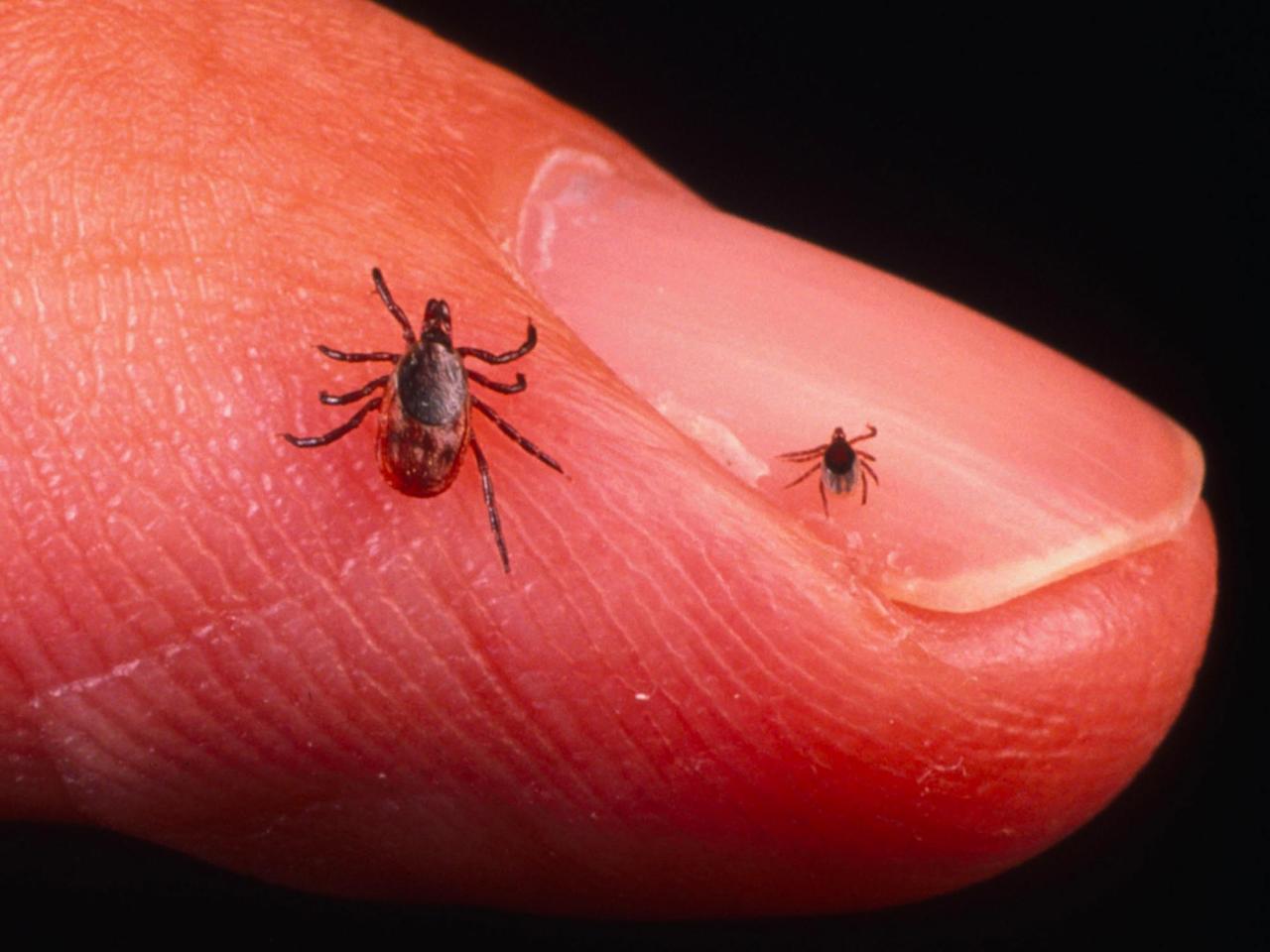
You Probably Wont See The Tick That Infects You
After hatching out of an egg, the deer tick passes through three stages: larva, nymph, and adult. At each stage, the tick must find a new host and feed. Since when they hatch they are uninfected by Borrelia, larva cant cause Lyme disease. And adult ticks are easy to feel and spot when they crawl on you, so theyre also unlikely to latch on and pass along the disease. The really dangerous vectors are the nymphs, about 15 percent of which carry Borrelia and which are so tiny less than a tenth of an inch long, about the size of a poppy seed that its very difficult to see them. Most people who come down with Lyme disease dont notice that theyve been bitten.
Tick nymphs are active in the spring, lurking in tall grass or shrubbery, waiting for a host to drop down onto. Since this time of year is also when human beings are most likely to be frolicking amid the greenery with lots of tasty exposed flesh, Lyme disease cases spike around now.
Where Are We Now
As Julia Belluz reported at Vox, Lyme cases doubled since 1991, spread by an increased number of infected ticks. Its now the most common vector-borne disease in the United States. And climate change seems to be partly to blame: As temperatures warm, a greater proportion of the US becomes hospitable to the ticks. Overall, vector-spread diseases like chikungunya, Zika, and West Nile are spreading faster than ever.
A safe and effective Lyme vaccine is desperately needed, Lise Nigrovic, a pediatric Lyme disease researcher at Boston Childrens Hospital, says in an email. Yet if you wanted to protect yourself with a Lyme disease vaccine, you couldnt get one. As Belluz explained, prevention efforts currently focus on avoiding tick bites. That means covering up exposed skin when spending time in wooded areas, using insect repellent, and checking your body for ticks after youve spent time outdoors in tick-laden areas.
WBUR in Boston reports there have been some small efforts to revive LYMErix , but the pharmaceutical industry has lost interest in it, and grassroots efforts have gone unfunded. The Lyme vaccine for dogs works in a similar manner to LYMErix. But while it does help control the spread of the disease, it doesnt make up for the lack of a vaccine in humans.
The loss of LYMErix represents the loss of a powerful tool for Lyme disease prevention, the authors of the Epidemiology and Infection article state.
Also Check: Signs And Symptoms Of Lyme Disease In Child
The Lyme Vaccine Can Cause Lyme Disease Symptoms
Holistic vet Dr Patricia Jordan urges pet owners to avoid the Lyme vaccine for their dogs. She claims the Lyme vaccine is more dangerous than Lyme disease:
There is no justification for taking this serious vaccination risk with our dogs. The Lyme vaccine is all risk and no benefit. There is a high chance of severe adverse events like a lifetime of non-treatable arthritis pain just for getting the jab in the first place.
And Dr Michael Garvey of the Animal Medical Center agrees. He says dogs can develop Lyme-like symptoms. And it can happen days or even weeks after vaccination.
Dr Jordan also explains that cells in the immune system can react with the antigens in the Lyme vaccine. This causes Lyme nephritis which is ironic, because Lyme nephritis is the reason your vet wants to give your dog the vaccine!
This is why none of the US veterinary schools list the Lyme vaccine as one of their recommended vaccines.
Why It Took So Long To Develop A New Vaccine For Lyme Disease
- Save Story
Save this story for later.
- Save Story
Save this story for later.
In May, an article with the unprepossessing title Detecting Borrelia Spirochetes: A Case Study with Validation Among Autopsy Specimens was published in the medical journal Frontiers in Neurology. The deceased person in question was a sixty-nine-year-old woman who suffered from severe cognitive impairment. Fifteen years before her death she had been treated for Lyme disease, the most prevalent tick-borne illness in the United States, and was thought to have fully recovered. Yet, when her brain and spinal-cord tissue were examined, researchers found intact Borrelia spirochetes, the bacteria responsible for Lyme. If the womans cognitive decline did result from Lyme diseasewhich the paper suggested was a strong possibilitythen it was further evidence that the illness could persist and wreak havoc long after a tick bite, and long after treatment.
It was the first time an F.D.A.-licensed vaccine was removed because of a concerted public-opinion campaign, even as the number of infections were rising. People say, Why cant I do for myself what I can do for my dog? Well that, you know, is thanks to the people who brought down LYMErix, Mark Klempner, a professor of medicine at the University of Massachusetts and one of the vaccines developers, told me. It was a great disappointment to have worked all those years and feel successful at the end of it, only to see it pulled. It was a tragedy.
Recommended Reading: Lyme Disease Test Covered By Insurance
Why Lyme Disease Is Hard To Treat
Lyme disease, which is caused by Borrelia burgdorferi bacteria, is a tick-borne illness. It doesnt circulate in the blood like many bacterial infections.
Instead, it hides in your bodys tissues. That makes it more difficult to detect and harder to treat.
If an infected tick bites you, you may begin to show signs of Lyme disease in one to four weeks.
Primary Lyme disease is what most of us are familiar with, May said. The symptoms include fever, fatigue, body and joint aches, and in some cases, the characteristic bulls-eye rash where red rings appear around the site of the tick bite.
If caught early, Lyme disease can be treated with antibiotics.
If its not caught, untreated Lyme disease may progress and become a bit more serious.
Secondary Lyme disease can include swollen or painful joints, extreme fatigue, numbness, headaches, muscle weakness, conjunctivitis, poor memory, confusion, and heart palpitations, May said.
If its still not diagnosed at this stage, the bacteria may invade the central nervous symptom and cause problems, such as mental status changes and effects on mood, sleep, and memory, May said.
Loss of muscle control, tics, a lack of coordination, and potentially deadly heart complications are also possible, she added.
Today, 90 percent of cases of Lyme disease are reported in just 14 states, according to the CDC. The majority of these states are in the Middle Atlantic region and New England.
Facts On Human Vaccination

There is a race to bring a new human Lyme disease vaccine to market, with some vaccines already in advanced stages of development.4, 5 A previous Lyme disease vaccine for humans had been approved for use in the U.S., but was removed from the market. The observation that this vaccine was unsuccessful due to lack of sales is often misused as an argument against canine Lyme disease vaccination.
The University of Pennsylvanias world renowned vaccinologist, Stanley Plotkin, described the human vaccine situation in 2011: A vaccine against Lyme disease was licensed in the United States in 1998 but was subsequently removed from the market because of lack of sales. I believe that the poor acceptance of the vaccine was based on tepid recommendations by the CDC, undocumented and probably nonexistent safety issues, and insufficient education of physiciansThe fact that there is no vaccine for an infection causing 20,000 annual cases is an egregious failure of public health.6
Also Check: Lyme Vaccine For Dogs How Often
Treatment For Other Forms Of Lyme Disease
People with other forms of disseminated Lyme disease may require longer courses of antibiotics or intravenous treatment with antibiotics such as ceftriaxone. For more information about treating other forms of Lyme disease, see:
The National Institutes of Health has funded several studies on the treatment of Lyme disease that show most people recover within a few weeks of completing a course of oral antibiotics when treated soon after symptom onset. In a small percentage of cases, symptoms such as fatigue and myalgia can last for more than 6 months. This condition is known as post-treatment Lyme disease syndrome , although it is also sometimes called chronic Lyme disease. For details on research into chronic Lyme disease and long-term treatment trials sponsored by NIH, visit the visit the National Institutes of Health Lyme Disease web siteexternal icon.
A Vaccines Cautionary Tale
In 1998, the FDA approved a Lyme vaccine composed of protein antigens from the surface of the Lyme bacteria, Borrelia burgdorferi.
A vaccine works by introducing proteins from the disease-causing agent into the body to trigger the bodys immune response, which includes making antibodies against bacterial proteins. Antibodies have been used to prevent and treat infectious diseases for over a century. In the case of the Lyme vaccine, it can take many months for the body to build up the necessary level of immunity to prevent infection. It also means that some of the antibodies induced by the vaccine can have off-target effects, or side effects.
The Lyme vaccine, known as LYMErix, largely reduced infections but was withdrawn from the market after three years because of limitations and controversy.
LYMErix needed to be administered by multiple injections over a year before immunity developed. Uncertainty about the length of immunity from the vaccine also raised questions of whether a booster shot would be regularly needed. Further, publicity about side effects such as arthritis, reported by some who had been vaccinated, contributed to its decline in popularity.
Today, a French biotech company, in collaboration with Pfizer, is attempting to develop a Lyme vaccine it is currently in clinical trials.
Recommended Reading: Lyme Disease And Multiple Sclerosis
Four Safe And Effective Vaccines
Compared to humans, dogs are lucky. There are currently 4 extremely safe, effective, and reliable vaccines7-9 on the market that have all been through the United States Drug Administration licensing requirements for safety, efficacy, purity, and potency.
All available canine Lyme disease vaccines produce borreliacidal antibodies in the dog in response to vaccinal outer surface protein A . These antibodies work in the ticks gut to bind the bacteria during the blood meal, sterilizing the gut of the tick and preventing transmission of bacteria into the dog.
OspC is the main immunogenic protein exhibited by Borrelia in the ticks salivary glands and in the dogs body during natural infection. Three of the current Lyme disease vaccines contain 2 strains of inactivated Borrelia isolates1 OspA producing strain and 1 unique OspC producing strain, which perhaps adds an additional layer of protection.