Why It Took So Long To Develop A New Vaccine For Lyme Disease
- Save Story
Save this story for later.
- Save Story
Save this story for later.
In May, an article with the unprepossessing title Detecting Borrelia Spirochetes: A Case Study with Validation Among Autopsy Specimens was published in the medical journal Frontiers in Neurology. The deceased person in question was a sixty-nine-year-old woman who suffered from severe cognitive impairment. Fifteen years before her death she had been treated for Lyme disease, the most prevalent tick-borne illness in the United States, and was thought to have fully recovered. Yet, when her brain and spinal-cord tissue were examined, researchers found intact Borrelia spirochetes, the bacteria responsible for Lyme. If the womans cognitive decline did result from Lyme diseasewhich the paper suggested was a strong possibilitythen it was further evidence that the illness could persist and wreak havoc long after a tick bite, and long after treatment.
It was the first time an F.D.A.-licensed vaccine was removed because of a concerted public-opinion campaign, even as the number of infections were rising. People say, Why cant I do for myself what I can do for my dog? Well that, you know, is thanks to the people who brought down LYMErix, Mark Klempner, a professor of medicine at the University of Massachusetts and one of the vaccines developers, told me. It was a great disappointment to have worked all those years and feel successful at the end of it, only to see it pulled. It was a tragedy.
Why Theres Still No Lyme Vaccine For Humans
False starts and fear have kept such a drug off the market for two decades.
Jessica Reeder worries about ticks and the diseases they carry. She had Lyme disease, her brother had Lyme disease, and every fall her children come home from school with notes reminding parents to do a nightly tick check. Even in Philadelphia, where Reeder lives, the bloodsuckers lurk in the woods around the playground. Her family used to go tent camping with friends, but they stopped after a few people in the group contracted Lyme.
It just got to the point where people were too stressed out about it, she says.
Reeder made sure that her shih tzu, Rory, got the canine Lyme vaccine, but she must protect the human members of her family the old-fashioned way: bug spray, long pants tucked into socks, and frequent tick checks. Theres no Lyme vaccine on the market for humans.
Valneva, a French biotech company focused on developing vaccines for infectious diseases, hopes to change that. Six years ago, the company began working on a vaccine against Lyme disease, which is now part of phase-two clinical trials in the United States and Europe.
Valnevas Lyme vaccine isnt the first designed for people. Twenty years ago, Reeder could have been immunized. From 1999 to 2002, SmithKline Beechamnow GlaxoSmithKlinesold a Lyme vaccine called LYMErix. But the company pulled LYMErix off the market after public backlash and a spate of lawsuits.
Risk Communication And Policy Implications
Risk communication represents the process of informing individual and collective decision-making by describing benefits as well as risks. As illustrated by the case of Lyme vaccine, the issues involved often become very complex and require an in-depth understanding to accurately access risk. However, the public attention span is typically short. The media often serve as the only channel through which the general public obtains its health information.
In the aftermath of the LYMErix market withdrawal, we must look for lessons learned. The vaccine developers believed they developed a safe and effective vaccine to prevent the most common tick-borne infection in the United States. Even available post-market surveillance failed to demonstrate convincing harm from the LYMErix vaccine. After review of available data, the FDA found insufficient evidence to support a causal relationship between the reported adverse effects and the vaccine and continued to permit use of the vaccine. However, the public’s perception of potential risks, heavily influenced by the negative press coverage and limited awareness of the benefits of the vaccine, decreased consumer demand for the vaccine.
Don’t Miss: Lyme Disease And Chest Pain
House Orders Pentagon To Reveal If It Turned Ticks Into Biological Weapons
A newly developed vaccine could prevent Lyme disease in humans.
The inoculation against the tick-borne disease, being developed at the University of Massachusetts Medical School, received federal approval last year and is now part of an initial clinical trial, Field & Stream reported.
Work on the vaccine is being spearheaded by Dr. Mark Klempner, a member of the schools biologics program who has been working on a Lyme cure for a decade, the outlet said.
A Lyme vaccine that used to be publicly available had been withdrawn and since we understood the mechanism of protection there, we were able to go right after the molecule that we thought would be protective, Klempner told the Patriot-News.
The vaccine is being tested on 66 patients in Lincoln, Nebraska, and could be available to the public in the the spring of 2023, the reports said.
According to a report in January by the Centers for Disease Control and Prevention, Lyme disease is more prevalent than previously thought with as many as 400,000 cases reported to the agency every year.
The disease is most commonly transferred to humans through bites from infected black-legged ticks, with the bites typically leaving a characteristic skin rash.
Lyme is rarely fatal but can be debilitating. Symptoms include fever, headaches and fatigue, and it can cause nerve damage, memory loss, and inflammation around the heart if left untreated.
Fda Approves Investigational New Drug Application For Lyme Prep A Pre
UMass Medical School Communications
The first human clinical trial of Lyme PrEP, a seasonal shot to prevent Lyme disease, has begun enrolling volunteers to evaluate the safety and pharmacology of the treatment. A pre-exposure prophylaxis developed at MassBiologics of UMass Medical School, Lyme PrEP uses a monoclonal antibody that protects against the disease. Approximately 60 volunteers will be enrolled in the Phase I trial.
This is an exciting milestone, said Mark Klempner, MD, executive vice chancellor for MassBiologics and professor of medicine. There is a real unmet public health need for the prevention of Lyme disease. There are more than 30 years of clinical experience that says a monoclonal antibody will be safe. The bigger question we hope to answer is how long will the antibody last in the circulation so when a tick bites you will be protected from Lyme disease.
Lyme disease is the most common vector-borne disease in the United States. It is caused by the bacterium Borrelia burgdorferi and rarely, Borrelia mayonii, and is transmitted to humans through the bite of infected blacklegged ticks. When an infected tick bites someone and begins to feed on their blood, Lyme-causing bacteria can slowly travel from the ticks gut to its salivary glands and then transfer the bacteria to the site of the bite.
Related media coverage:
Recommended Reading: Deer Tick Lyme Disease Treatment
Disease Transmission And Symptoms
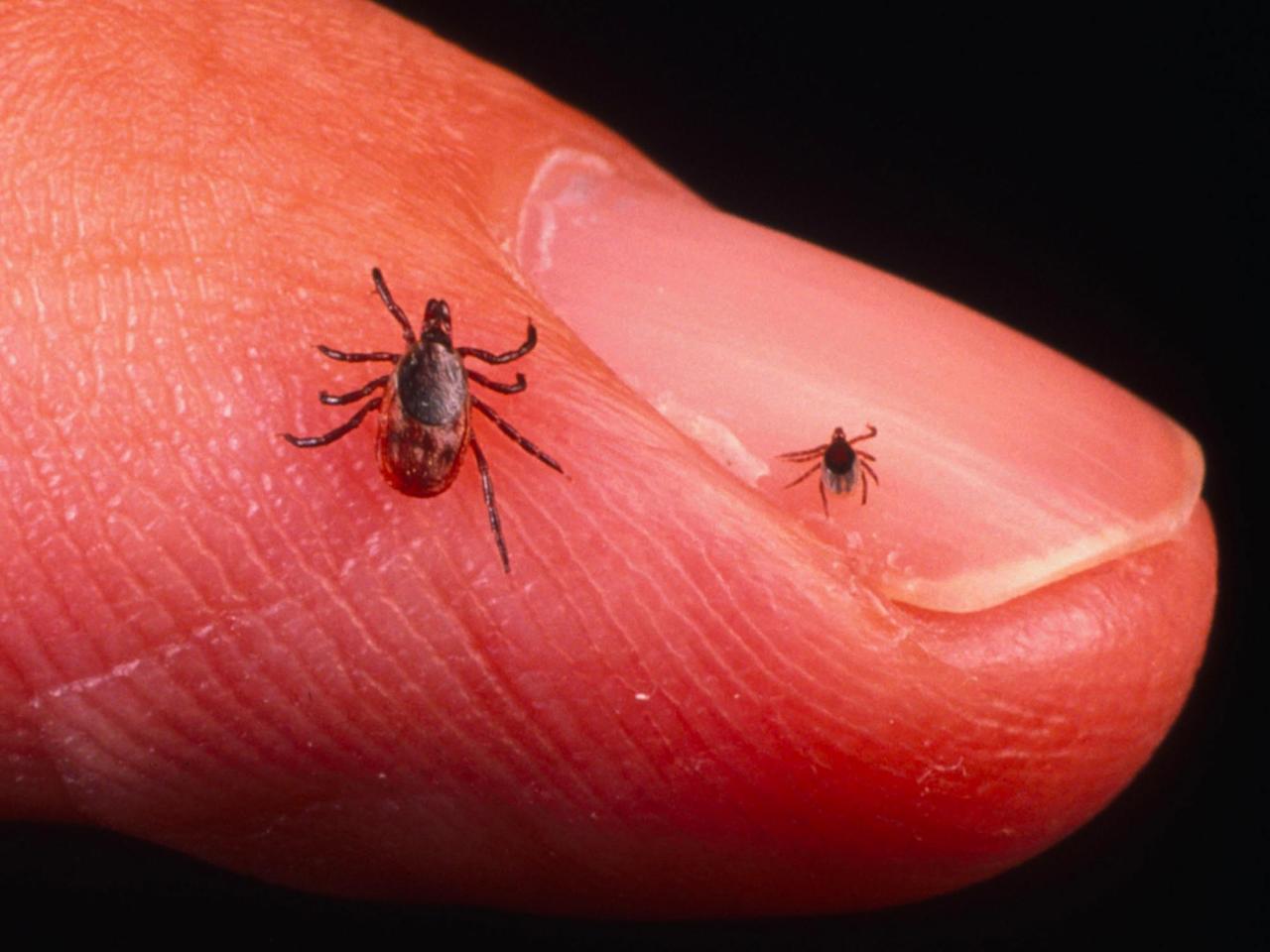
Lyme disease is caused by the Borrelia burgdorferi bacteria, passed to humans through the bite of infected black-legged ticks , which initially get the disease from mice. The transmission of the bacteria from an infected tick to a human can take hours often more than a day but the ticks small size makes them easy to overlook on the body, allowing time for this transmission to occur unimpeded. They are typically about the size of the head of a pin, and can bite without being noticed.
The most commonly known symptom of Lyme disease is the bulls-eye or target rash, which appears in most cases. The rash begins at the site of the tick bite between three and 30 days after exposure, and usually grows in size for several days. When Lyme disease is diagnosed, antibiotics are prescribed for treatment. If the disease is left untreated, other symptoms can develop in the weeks following exposure: additional rashes joint pain and swelling shooting pains dizziness and heart palpitations severe headaches and loss of muscle tone in the face .
If the disease remains untreated beyond this point, arthritis can develop. This occurs in about 60% of patients whose infections are not treated, and can cause swelling and severe pain in the joints. In addition, as many as 5% of patients whose Lyme infections are not treated develop chronic neurological problems. These can occur months or even years after transmission.
The First Meeting Of The Fda Lyme Disease Vaccine Advisory Panel: May 1998
Editorial reviews on the demise of the Lyme disease vaccine have been written,,,,,, two of which, discuss risk communication and policy implications. We agree that scientific evidence and best patient care practices should guide the ethics of Lyme disease activism. However, we also acknowledge that unclear, sometimes contradictory scientific terminology may have led to confusion that drove health care professionals vaccine hesitancy and subsequent public skepticism.
You May Like: Oxygen Therapy For Lyme Disease
Researchers Develop Lyme Disease Vaccine That Targets Tick Saliva Prevents Feeding
Yale University researchers have developed an mRNA vaccine that provides partial protection against Lyme diseasethe most common Tick-borne human illness in the United States.
First published on November 17 in the journal Science Translational Medicine, the novel vaccine developed and tested in guinea pigs offers protection against infection by the bacterium that causes Lyme disease. It may also combat other tick-borne diseases.
Specific ticks studied included Ixodes scapularimore commonly known as a deer tickwhich carries a Lyme-disease-causing bacteria called Borrelia burgdorferi.
Rather than target specific viruses or bacteria that can cause disease, this new vaccine is designed to target tick salivary proteins that are released through open wounds. So instead of triggering an immune response against a particular pathogen, this vaccine limits the amount of time that ticks have to feed upon and infect a host.
Researchers said the vaccine mirrors the same mRNA technology that has proved effective against COVID-19.
While a minimum of 40,000 Lyme disease cases exist across the United States, researchers believe the actual infection rate could be 10 times greater. Meanwhile, other tick-borne diseases have also spread domestically.
Yale researchers collaborated with the University of Pennsylvania to analyze mRNA bits that produce all 19 of the saliva proteins.
Closing In On A Safe And Effective Human Vaccine For Lyme Disease
One of VCUs most successful and revenue-generating inventors is on track to develop a vaccine against Lyme disease in humans as well as a new diagnostic test.
The human vaccine work builds on a USDA-approved Lyme disease vaccine originally developed for use in dogs. The canine vaccine is marketed under the brand name Vanguard® crLyme and produced and distributed by the animal health company Zoetis. It entered the veterinary market in 2016 and is now the most widely used Lyme disease vaccine in North America.
The canine vaccine has taken the veterinary market by storm, says Magdalena Morgan, Ph.D., director of licensing at VCU Innovation Gateway. Its considered the best of several such vaccines available, thanks to the vaccines capability to fight different strains of the disease. Its very versatile, she says.
While work on a human Lyme disease vaccine continues in the lab, we have the advantage of having taken the vaccine to a higher animal model and have demonstrated the efficacy and safety there, says vaccine developer Richard T. Marconi, Ph.D., a professor in the Department of Microbiology and Immunology. Using the same technological approach that was employed to develop the canine vaccine, Marconis lab is now developing a potential point-of-care test for Lyme disease.
You May Like: Initial Signs Of Lyme Disease
Lyme Disease Vaccine Found To Be Safe And Effective In Clinical Trial
A new Lyme disease vaccine is on the way
A vaccine against Lyme disease has been shown to be safe and effective in a clinical trial and could be available by 2025.
Tens of thousands of people in the US and Europe are diagnosed each year with Lyme disease, which is transmitted by tick bites and can cause lifelong health problems like joint and nerve pain if it isnt treated early.
French company Valneva has developed a vaccine that works by stopping Lyme-causing bacteria in ticks from passing into peoples bloodstreams
Continue reading
If You Have Lyme Disease You May Have Another Illness Too
Ticks have been called the dirty syringes of the animal kingdom, swapping infected blood indistriminately amongst their hosts. So if Lyme disease were the only thing we could get from these vermin, wed be lucky. And were not. There are a half dozen or more other tick-born pathogens that can get passed on along with Borrelia, including Babesia microti and Anaplasma phagocytophila . When someone is diagnosed with Lyme disease they should at least be tested for other pathogens, Telford says.
While waiting for new treatments to come along, your best bet is to take some simple precautions to avoid encountering Borrelia in the first place: wear long-sleeved clothes when in tick habitat, apply DEET insect repellent, and check yourself for embedded ticks. And if worse comes to worse and you come down with symptoms, talk to your doctor about getting tested. Antibiotics like doxycycline are highly effective against the disease even in its more serious later stages.
After all, says Telford, Lyme disease is a drag, but you shouldnt let the fear of it ruin your life: People should enjoy the outdoors, he says.
Don’t Miss: Home Remedies For Lyme Disease
Gene Editing To Stop Lyme Disease: Caution Is Warranted
Theres a lot of worry about Lyme disease, to the degree that some parents are telling children they shouldnt play outside or limiting hours, Nigrovic said. Theyre behaving differently because of that worry. So vaccines are attractive.
The Food and Drug Administration gave VLA15 fast-track designation in July 2017. Valneva completed initial safety studies in a Phase 2 clinical trial and, according to a company press release, VLA15 had no associated safety concerns. The company is now working to determine the dose. Based on current estimates, Lingelbach said Valneva plans to test the vaccine in a clinical trial of at least 15,000 people, and it should be available in four or five years.
The FDA approval process is long. But Telford said it may be easier for VLA15 than it was for LYMErix, given how many more people today know about Lyme disease and may enroll in trials.
Finding people is going to be a cinch compared to what it was like in the 1990s, he said. I dont see why they couldnt have it benefiting people by 2024.
A different approach, being developed by the University of Massachusetts Medical School, is called Lyme pre-exposure prophylaxis . Unlike a vaccine, Lyme PrEP delivers a single defensive antibody, said Dr. Mark Klempner, the executive vice chancellor for MassBiologics and a professor of medicine at the university, who is leading efforts to develop the product.
Vaccine Licensure Recommendation And Initial Use

In response to growing reports of Lyme disease cases in the United States from 1982 to 1996, the number of reported cases increased by 32 times SmithKline Beecham developed LYMERix, which was licensed in 1998. The licensed product was a recombinant vaccine containing an outer surface protein from theBorrelia burgdorferi bacteria. Before licensure, 6,478 people received 18,047 doses of the vaccine during clinical testing. The most common adverse events noted within 30 days of receiving at least one dose of the vaccine included pain or reaction at the injection site, joint pain, muscle pain, and headache. Of these, only pain and reactions at the injection site occurred much more frequently in the vaccine recipients than in those who received a placebo.
The efficacy trial for the vaccine showed that it was 78% effective in preventing Lyme disease after all three doses were given. It was also shown to be 100% effective at preventing asymptomatic cases, where an individual would get the disease and develop antibodies against it but never develop any symptoms.
Based on the clinical trial data, the vaccine was given a permissive recommendation by the Advisory Committee on Immunization Practices. A permissive recommendation means a vaccine is not added to the childhood or adult immunization schedules, like vaccines against common childhood diseases . Instead, the vaccine is considered for use only in individuals or groups with specific risk factors for a disease.
Recommended Reading: Should I Be Tested For Lyme Disease
Vaccines: Our Greatest Hope In Defeating Aids Ebola Tb And Other Infectious Diseases
Safety concerns began to emerge soon after people began getting LYMErix. Some recipients began to report joint pain and other effects that they attributed to the vaccine. Within a year of the vaccines approval, a class action lawsuit was filed against SmithKline Beecham on behalf of 121 people. Although the suit was eventually settled it provided no compensation to the plaintiffs publicity about it dampened interest in the vaccine.
Meanwhile, growing distrust of vaccines may have compounded LYMErixs struggles.
When a 1998 report in the Lancet identified a purported link between the common MMR vaccine and autism, the study since retracted and considered fraudulent sparked a spike in worry about the safety of vaccines. In October 1999, a new vaccine against rotavirus, the most common cause of diarrhea in infants and young children, was withdrawn from the market after its use was linked to a higher than expected risk of a potentially deadly bowel blockage known as intussusception.
An advisory panel convened in 2001 by the FDA did not find any evidence that LYMErix caused arthritis in humans or an unexpected number of adverse events. But growing suspicion of vaccines in general and publicity about possible side effects had already taken a toll on LYMErixs uptake. SmithKline Beecham projected sales of only 10,000 doses for 2002.
The company withdrew LYMErix in February 2002, citing poor market performance.