Who Can Participate In A Clinical Trial
All clinical trials have guidelines about who can participate, called inclusion criteria. The plan will also have factors that disqualify a person from taking part, called exclusion criteria. These standards include parameters such as age, gender, type and stage of disease, previous treatment history, and other medical conditions. Only those people that meet all of the inclusion criteria and none of the exclusion criteria can participate in a clinical trial.
The selection criteria ensure that researchers are studying the right people to help find answers to important medical questions.
If you are interested in volunteering, first find out if you are right for that clinical trial and if the clinical trial is right for you.
The safety and well-being of study participants are top priorities for the entire research team. If you choose to participate in a clinical trial, your health will be closely monitored, and your care will be adjusted if the research team thinks its in your best interest. If you decide at any point to end your study participation, you may do so in consultation with the PI and research team.
Existing Lyme Disease Treatments Do Not Always Work
Most people diagnosed with Lyme are treated with an oral antibiotic, doxycycline however, as many as 20% of those treated do not get relief. It appears the bacteria have a tolerance to the drug. When bacteria is threatened, it morphs into a different form that cant be affected by treatment. When the treatment is over, the bacteria changes back to its active form. Sneaky little bugger, huh? This possibly explains how some people can stay sick with Lyme for so long, even after repeat treatment.
Monoarthritis Risk Stratification In Lyme Disease
In Lyme disease endemic areas, a noninvasive clinical prediction model to distinguish septic arthritis from Lyme arthritis would be useful, especially in children. A new study suggests that in highly selected patients, such prediction might be possible. But failure to recognize even a few cases of septic arthritis could have devastating consequences.
You May Like: Lyme Disease Chain Of Infection
About The Steven And Alexandra Cohen Foundation
The Steven & Alexandra Cohen Foundation is committed to inspiring philanthropy and community service by creating awareness, offering guidance, and leading by example to show the world what giving can do. The Foundations grants support nonprofit organizations based in the United States that either help people in need or solve complex problems. The Foundation also spearheads grassroots campaigns to encourage others to give. For more information, visit www.steveandalex.org.
Azlocillin Comes Out On Top
The drug, which is not on the market, was tested in mouse models of Lyme disease at seven-day, 14-day and 21-day intervals and found to eliminate the infection. For the first time, azlocillin was also shown to be effective in killing drug-tolerant forms of B. burgdorferi in lab dishes, indicating that it may work as a therapy for lingering symptoms of Lyme disease.
Pothineni and Rajadas have patented the compound for the treatment of Lyme disease and are working with a company to develop an oral form of the drug. Researchers plan to conduct a clinical trial.
Rajadas is also a professor of bioengineering and therapeutic sciences at the University of California-San Francisco.
Other Stanford co-authors are Hari-Hara S. K. Potula, PhD, senior research scientist postdoctoral scholars Aditya Ambati, PhD, and Venkata Mallajosyula, PhD senior research scientist Mohammed Inayathullah, PhD and intern Mohamed Sohail Ahmed.
A researcher at Loyola College in India also contributed to the work.
The study was funded by the Bay Area Lyme Foundation and Laurel STEM Fund.
- Tracie White
Recommended Reading: Dog Test For Lyme Disease
Fda Approves Investigational New Drug Application For Lyme Prep A Pre
UMass Medical School Communications
The first human clinical trial of Lyme PrEP, a seasonal shot to prevent Lyme disease, has begun enrolling volunteers to evaluate the safety and pharmacology of the treatment. A pre-exposure prophylaxis developed at MassBiologics of UMass Medical School, Lyme PrEP uses a monoclonal antibody that protects against the disease. Approximately 60 volunteers will be enrolled in the Phase I trial.
This is an exciting milestone, said Mark Klempner, MD, executive vice chancellor for MassBiologics and professor of medicine. There is a real unmet public health need for the prevention of Lyme disease. There are more than 30 years of clinical experience that says a monoclonal antibody will be safe. The bigger question we hope to answer is how long will the antibody last in the circulation so when a tick bites you will be protected from Lyme disease.
Lyme disease is the most common vector-borne disease in the United States. It is caused by the bacterium Borrelia burgdorferi and rarely, Borrelia mayonii, and is transmitted to humans through the bite of infected blacklegged ticks. When an infected tick bites someone and begins to feed on their blood, Lyme-causing bacteria can slowly travel from the ticks gut to its salivary glands and then transfer the bacteria to the site of the bite.
Related media coverage:
Rural And Urban Differences
The infographic reveals key differences in prevalence of Lyme disease when comparing rural and urban areas. From 2016 to 2021, claim lines with Lyme disease diagnoses increased 60 percent in rural areas and 19 percent in urban areas, with diagnoses peaking nationally in June and July of each year.
During these summer months, rural areas, on average, had a greater share of claim lines associated with Lyme disease diagnoses than urban areas. From November to April, however, claim lines with Lyme disease diagnoses occurred more often in urban than rural areas.
Also Check: Can Lyme Disease Cause Headaches
You May Like: Can Lyme Disease Cause High Blood Pressure
New Drug Kills All Forms Of The Lyme Bacteria
Rajadas and Pothineni put together a team to study new drugs that could fight Lyme disease more effectively. Their study was recently published in Scientific Reports. They took thousands of existing drugs and chemical compounds and mixed them with Lyme. As results seemed promising, they created second and third tests to recheck those that showed positive results. Through this process, the team found a drug called azlocillin.
Azlocillin was previously approved by the FDA for the treatment of alcohol abuse disorder. Azlocillin appears to kill the morphed forms of the bacteria as well as the active. This could lead to a significant decrease in the need for extended treatment for Lyme disease. We are pleased to share that the treatment is now moving into human clinical trials.
An effective treatment for Lyme disease and the bacteria that cause it would be an astonishing scientific breakthrough. It could ease hundreds of thousands of people from the suffering that is associated with the disease. At Mosquito Squad of Southeastern Massachusetts, we are excited to share this promising news. Even still, we must remind you of one thing: Ticks are already out this year.
Clinical Trials Network Coordinating Center For Lyme And Other Tick
CTN launched a new website and welcomes your ideas on novel treatment approaches in the field of Lyme and the Tick-borne diseases.
In recognition of a severe unmet need, the Steven & Alexandra Cohen Foundations grant to Columbia University investigators establishes the first National Clinical Trials Network Coordinating Center for Lyme and other Tick-borne Diseases. Under the leadership of Dr. Brian Fallon, this Clinical Trials Coordinating Center aims to propel national research toward the goal of identifying more effective treatments for patients with Lyme and other tick-borne diseases.
Providing oversight to ensure that the highest-quality clinical trials are undertaken, the CTN Coordinating Center at Columbia will provide funding support for pilot studies, assist investigators in protocol design and statistical research planning, establish and manage a national case registry, and create a data management system for multi-site clinical trials. The clinical trials network will be a powerful engine to drive large-scale clinical trials and potentially transformational early stage research.
Invitation for input and collaboration
The CTNCC welcomes input from the public, community clinicians, and academics regarding novel treatment approaches that we should review and consider. If you would like to be placed on our mailing list to be kept up to date on the Clinical Trial Network announcements, please fill out our Contact Us form or email us at .
Also Check: Do All Ticks Have Lyme Disease
Growing Need For Lyme Disease Experts And Research
The estimated incidence of Lyme disease in the United States has been steadily rising, to approximately 476,000 new cases annually. Despite antibiotic treatment, approximately 10-20% of these new infections will lead to distressing and potentially disabling symptoms, such as severe fatigue, joint and muscle pain, neurologic symptoms, and cognitive problems that may last for months or years.
There is limited expertise in treating the acute and chronic aspects of tick-borne diseases, making it difficult for patients to find high-quality, specialized care. In addition, little research has been done to determine which treatments are most effective for persistent symptoms, which can interfere with daily activities and work.
Like COVID-19 long-haulers, many people with tick-borne diseases were completely well until their infection precipitated a cascade of chronic, multi-system effects, says Brian Fallon, MD, director of the Cohen Center for Health and Recovery from Tick-Borne Diseases. A comprehensive evaluation takes time. In addition, the cost of care for patients with chronic symptoms can be prohibitive, requiring multiple visits to physicians who may not be aware of the latest research on tick-borne illnesses. Our center will be the first to address all of these issues by offering access to affordable care with experienced physicians while at the same time integrating research and physician training into our clinical model.
Biorepositories And Research Cohorts
Well-characterized samples are an essential tool to help researchers develop and validate new diagnostic tests and to better understand the complexities of LD. Well-characterized sample sets can benefit medical providers, test developers, and the public at risk for LD . It is critical that sample users understand the criteria used to enroll participants, how samples were collected and stored, and what additional clinical and testing data may be available. Additional benefits can be realized when multiple sample users are using the same well-characterized sample sets. Current sample sets available for researchers include the CDC Lyme Serum Repository , the Lyme Disease Biobank , and samples from the Studies of Lyme Immunology and Clinical Events at Johns Hopkins University School of Medicine. Additionally, some investigators also have their own sample collections with, in some cases, blood samples, skin biopsy specimens and synovial fluid which form the basis for collaborative studies .
Lyme Disease Biobank
You May Like: What Herbs Kill Lyme Disease
Also Check: Can Lyme Disease Cause Afib
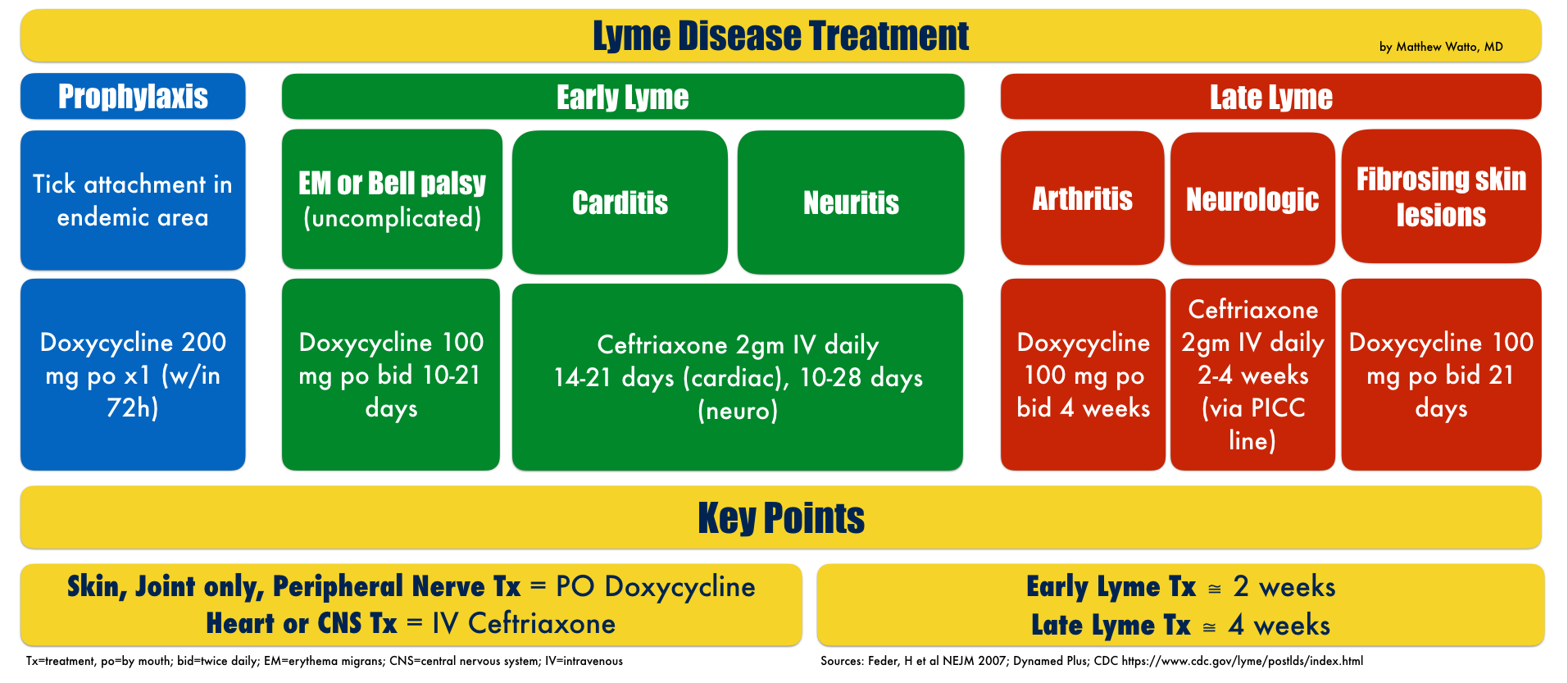
Potential Treatment For Lyme Disease Kills Bacteria That May Cause Lingering Symptoms Study Finds
Screening thousands of drugs, Stanford scientists determined that in mice, azlocillin, an antibiotic approved by the Food and Drug Administration, eliminated the bacteria that causes Lyme disease.
Deer ticks are vectors of Borrelia burgdorferi, the bacteria that causes Lyme disease.Scott Bauer/USDA Agricultural Research Service
For decades, the routine treatment for Lyme disease has been standard antibiotics, which usually kill off the infection. But for up to 20% of people with the tick-borne illness, the antibiotics dont work, and lingering symptoms of muscle pain, fatigue and cognitive impairment can continue for years sometimes indefinitely.
A new Stanford Medicine study in lab dishes and mice provides evidence that the drug azlocillin completely kills off the disease-causing bacteria Borrelia burgdorferi at the onset of the illness. The study suggests it could also be effective for treating patients infected with drug-tolerant bacteria that may cause lingering symptoms.
This compound is just amazing, said Jayakumar Rajadas, PhD, assistant professor of medicine and director of the Biomaterials and Advanced Drug Delivery Laboratory at the Stanford School of Medicine. It clears the infection without a lot of side effects. We are hoping to repurpose it as an oral treatment for Lyme disease. Rajadas is the senior author of the study, which was published online March 2 in Scientific Reports. The lead author is research associate Venkata Raveendra Pothineni, PhD.
Groundbreaking Johns Hopkins Studies
A 2006 publication by Johns Hopkins on the safety and enduring positive effects of a single dose of psilocybin is widely considered a landmark study sparked a renewal of psychedelic research worldwide.
Since then, they have further groundbreaking studies in more than 60 peer-reviewed articles in respected scientific journals.
This makes Johns Hopkins the leading psychedelic research institution in the U.S., and among the few leading groups worldwide.
Candidates for trials
In partnership with Columbia University, CPCR researchers recently launched a new survey study on how psychedelic experiences can impact romantic relationships. Couples are eligible to participate if both members of the couple have used a psychedelic together, or only one member of the couple used a psychedelic during the time they were in their romantic relationship.
Don’t Miss: How Soon Should You Treat Lyme Disease
Can Late Stage Lyme Be Treated
“Late-stage Lyme disease can be cured with antibiotics for 28 days. Arthritis and neurological disorders are the common symptoms of late-stage Lyme disease and are treated differently than in the early stages. Help clinical trial experts find the next breakthrough in treating Lyme disease by participating in one of the clinical trials within your location. Go to Power, and match with the best clinical trials that suit your current condition.” – Anonymous Online Contributor
Recent Progress In Lyme Disease And Remaining Challenges
Lyme disease is the most common vector-borne disease in the United States with an estimated 476,000 cases per year. While historically, the long-term impact of Lyme disease on patients has been controversial, mounting evidence supports the idea that a substantial number of patients experience persistent symptoms following treatment. The research community has largely lacked the necessary funding to properly advance the scientific and clinical understanding of the disease, or to develop and evaluate innovative approaches for prevention, diagnosis, and treatment. Given the many outstanding questions raised into the diagnosis, clinical presentation and treatment of Lyme disease, and the underlying molecular mechanisms that trigger persistent disease, there is an urgent need for more support. This review article summarizes progress over the past 5 years in our understanding of Lyme and tick-borne diseases in the United States and highlights remaining challenges.
Don’t Miss: What Percentage Of Deer Ticks Carry Lyme Disease
Can You Get Lyme Disease Without A Tick Bite
“Lyme disease can only be transmitted when an infected tick bites you. It means that you cannot be infected even by close contact with a person who currently has the infection. Clinical trials aim to develop a new drug or vaccine for Lyme disease. These trials require participants subject to some requirements to help with the studies. If you are interested in participating in one of the clinical trials, visit Power to help you match with a clinical trial according to your condition and location.” – Anonymous Online Contributor
Does Cbd Help Lyme Disease
“CBD is known for its many beneficial properties, including its anti-bacterial benefit. In addition, several symptoms of Lyme disease like fever, pain, stress, and anxiety are said to be reduced by CBD. Clinical trials have a long history of researching both CBD and Lyme disease, and one can even participate in these trials. Use the website of Power to easily find a clinical trial that is tailored to your current condition, treatment plans” – Anonymous Online Contributor
Don’t Miss: Does Lyme Disease Make Your Joints Hurt
Hhs Federal Research Updates On Lyme Disease Diagnostics
media iconLow Resolution VideoLinks with this icon indicate that you are leaving the CDC website.
- The Centers for Disease Control and Prevention cannot attest to the accuracy of a non-federal website.
- Linking to a non-federal website does not constitute an endorsement by CDC or any of its employees of the sponsors or the information and products presented on the website.
- You will be subject to the destination websiteâs privacy policy when you follow the link.
- CDC is not responsible for Section 508 compliance on other federal or private website.
You May Like: Can You Heal From Lyme Disease
How Do Researchers Implement A Clinical Research Idea
Everything that happens in each phase of a clinical trial follows a plan detailed in a study plan, also referred to as a protocol, which describes the following:
- Purpose of the research
- Goals of the research such as the type of information to be learned from the clinical trial
- Eligibility requirements defines the population of people that can participate in the clinical trial .
- Details about tests, procedures, and/or treatments
- How long the clinical trial will last
- Potential benefits to participants
- Risks to participants and how the research team will minimize the risks
Don’t Miss: Can Chronic Lyme Disease Be Cured
Why Is This So Important
According to the CDC, Lyme Disease is caused by infection with the bacterium Borrelia burgdorferi. Although most cases of Lyme disease can be cured with a 2- to 4-week course of oral antibiotics, patients can sometimes have symptoms of pain, fatigue, or difficulty thinking that last for more than 6 months after they finish treatment. This condition is called Post-Treatment Lyme Disease Syndrome .
It is not known why some patients experience PTLDS. Some experts believe that Borrelia burgdorferi can trigger an auto-immune response causing symptoms that last well after the infection itself is gone. CDC
No proven treatment to date for PTLDS
The CDC reports that unfortunately there is no proven treatment for PTLDS.
Although short-term antibiotic treatment is a proven treatment for early Lyme Disease, funded by the National Institutes of Health it has found that long-term outcomes are no better for patients who receive additional prolonged antibiotic treatment than for patients who receive a placebo. Long-term antibiotic treatment for Lyme disease has been associated with serious, sometimes deadly complications, as described in the links below.
Shania Twain/Instagram
Herpes Lyme Disease Ramsay Hunt Syndrome: What Happened To Justin Bieber
The diagnosis that Justin Bieber faced is not treated in 90% of cases
Justin Bieber recorded a sad video where his face is distorted and partially paralyzed. The singer told his fans that he suffers from borreliosis , which is transmitted by ticks. By the way, there are also borrelitosis-infected arthropods in the Moscow region and even in Moscow.
In addition, his doctors announced the diagnosis: Ramsay -Hunt syndrome.
Also Check: Can Lyme Disease Be Chronic
Read Also: Rocky Mountain Spotted Fever Vs Lyme Disease