Detection Of Dna: Nucleic Acid Amplification Tests
For infectious agents that are difficult to visualize or cultivate, nucleic acid amplification tests can identify specific pathogen DNA. Many pathogens are easily detected using NAATs, especially with high enhancement or enrichment methods when present. Many polymerase chain reaction assays have been geared to analyze small volumes of blood, plasma, or other sample types. In Lyme disease, outside the EM in skin, the infectious agent resides in very low numbers in tissue and body fluids, and standard PCR methods, as applied to B. burgdorferi detection, have suffered from poor sensitivity, especially in blood and cerebrospinal fluid . An illustrative strategy to increase PCR sensitivity without diminishing specificity is to start with a larger specimen volume and/or use target enrichment methods . It appears possible to detect B. burgdorferi in the blood with higher sensitivity in early Lyme disease. In addition, there are a variety of PCR-based assays in development that use unconventional signal detection or amplification methods , with the potential to achieve a limit of detection that is substantially lower than with standard PCR methods.
An additional hopeful point for future acute NAAT development is that some acute Lyme disease studies have documented that the number of organisms or their DNA in the blood may have been higher than appreciated .
You Do Not Usually Need Tests To Show That You Have Lyme Disease
In most cases, theres a clear sign of Lyme diseasea painless, spreading rash that often grows to look like a bulls eye. If you have this rash, and you recently had a tick bite or were in an area known for Lyme disease, you dont need a test. Instead, your doctor can just start treating you with antibiotics, as appropriate.
Interpreting The Igm Western Blot Test: The 1
If clinical symptoms and signs of Lyme disease have been present for more than 1 month, IgM reactivity alone should not be used to support the diagnosis, in view of the likelihood of a false-positive test result in this situation. This is called the 1-month rule in the diagnosis of Lyme disease.
In early localized infection, Western blot is only half as sensitive as ELISA testing. Since the overall sensitivity of a 2-step algorithm is equal to that of its least sensitive component, 2-tiered testing is not useful in early disease.
Although currently considered the most specific test for confirmation of Lyme disease, Western blot has limitations. It is technically and interpretively complex and is thus not universally available. The blots are scored by visual examination, compromising the reproducibility of the test, although densitometric blot analysis techniques and automated scanning and scoring attempt to address some of these limitations. Like the ELISA, Western blot can have false-positive results in healthy individuals without tick exposure, as nonspecific IgM immunoblots develop faint bands. This is because of cross-reaction between B burgdorferi antigens and antigens from other microorganisms. Around 50% of healthy adults show low-level serum IgG reactivity against the FlaB antigen, leading to false-positive results as well. In cases in which the Western blot result is indeterminate, other etiologies must be considered.
Also Check: What To Do If You Have Lyme Disease
Diagnostic Testing For Lyme Disease: Beware Of False Positives
Lyme disease is caused by the spirochete Borrelia burgdorferi and transmitted mainly via the tick vector Ixodes pacificus in British Columbia. Polymerase chain reaction testing finds Borrelia burgdorferi in only 1 in 200 ticks, whether they are collected from the wild or submitted by patients. This frequency is fiftyfold lower than in high-incidence areas of North America.
Early Lyme disease is a clinical diagnosis based on exposure to a tick bite followed by the characteristic erythema migrans rash within 4 weeks of exposure. Later in the course of infection serological testing is used. As with all other tests it is necessary to consider the validity of the test, including sensitivity and specificity.
Alternative laboratories have devised their own nonvalidated tests, which do not follow the two-step process. While these labs strive for high sensitivity they do not outperform reference labs in finding Lyme disease. Moreover, specificity as low as 43% is reported, indicating that 57% of results coming from such labs are false positives.
Advocates of alternative tests express concern about false negative results, so it must be noted that 99.5% of errors in alternative lab testing come from false positives and that they do not lower the false negative rate. Meanwhile, reference laboratories have vastly lower error rates and are working on ways to further improve test sensitivity.
hidden
The Clinical Diagnosis Of Lyme Disease
Misconception: Lyme disease is a clinical diagnosis that should be made based on a list of symptoms.
Science: No clinical features, except erythema migrans or possibly bilateral facial nerve palsyin the appropriate contextprovide sufficient specificity or positive predictive value. Laboratory confirmation is essential except with erythema migrans.
Read Also: Cvs Minute Clinic Lyme Disease Test
The Best Test For Lyme Disease
Lyme disease is a tick-borne illness spread by Lyme borreliaebacteria which includes, but is not limited to, Borrelia burgdorferi sensu stricto. The Centers for Disease Control and Prevention estimate that there are 400,000 cases of Lyme disease annually, making Lyme a serious public health concern that only stands to grow as the spread of ticks affects disease endemicity and seasonality.
One of the biggest challenges of fighting Lyme disease is providing patients with accurate diagnostic tests. Without access to the best tests for Lyme disease, its impossible to diagnose this treatable disease in a timely manner. When the disease isnt caught in time, it can spread throughout the body and cause chronic health problems that could otherwise be avoided with earlier detection and treatment.
Lyme Testing: Accurate When Used Appropriately
A 2016 systematic review that included 8 studies of CDC-recommended two tier test performance in Late Lyme showed Lyme antibody testing to be 99.4% sensitive and 99.3% specific** in North America.
In other words, out of 100 people with Late Lyme disease, 99-100 of them will test positive. Out of 100 people who may believe they have Late Lyme disease but do not, 99-100 of them will test negative.
Misinformation can convince patients to ignore or misinterpret negative tests to justify a false Chronic Lyme diagnosis.
Recommended Reading: What Type Of Doctors Treat Lyme Disease
How Much Does A Lyme Disease Test Cost
The cost of a Lyme disease test depends on what type of test is performed an ELISA test or a combination of ELISA and western blot tests.
Averagely, the cost for an ELISA test for Lyme disease is in the vicinity of $120 or more. If you include a confirmation western blot test, it can attract an additional $130 or more. More details on pricing can be referenced on our website.
Although an ELISA test might come back negative, it is best to combine it with a western blot test because not everyone shows the rash or bulls eye symptoms of Lyme disease.
After a successful diagnosis and treatment, it is equally important to do a follow-up test for Lyme disease years later to be sure that any similar flu-like symptoms are something else and not the same disease reoccurring.
Does The Time Of Year Matter In The Diagnosis Of Lyme Disease
Since Lyme disease is a tick-borne infection, the seasonality of the disease is linked tightly to the life cycle and behavior of ticks. May, June, July, and early August are the biggest risk months for acquiring first stage Lyme disease in the US. This is the feeding time for nymph and adolescent ticks which are difficult to see. A viral-like illness in those early summer months might be Lyme disease. There is also some transmission of Lyme disease via adult ticks in the fall and winter and throughout the year anytime the temperature is above 40 degrees, but to a lesser degree.
The risk of acute Lyme disease is more of a year-round risk in more temperate regions such as northern California and the pacific northwest. Later stage Lyme disease, however, can manifest at any time.
Also Check: Tick Bite Lyme Disease Rash
Serological Testing Of Csf For The Diagnosis Of Neurological Lyme Disease
Serological testing for neurological Lyme disease is based on demonstrating intrathecal synthesis of Borrelia-specific antibodies in CSF. For laboratory testing for neurological Lyme disease, separate IgG and IgM ViraChip® serology assays are performed on CSF and paired serum and the results compared.
CSF samples must be tested in parallel with a contemporaneous serum sample and protein and immunoglobulin levels compared between the two sample types to produce a meaningful result.
For necessary sample types and volumes see Sample types for Lyme disease testing
New Methods Are Needed
There is a lag period in most infections, and with B. burgdorferi, up to 3 weeks may be needed from infection to B-cell production of pathogen-specific antibody in sufficient quantity to be detectable by current serologic assays. Also immunoglobulin M reactivity is fraught with issues that affect utility. Culture of the pathogen, microbial nucleic acid, or protein may be detected in skin biopsies of erythema migrans before the development of a positive serology. However, culture is impractical for routine clinical use. It requires specialized media B. burgdorferi grows slowly, requiring weeks before it comes detectable. Although improvements in serologic assays are foreseeable, current assays utilize antigen targets identified before a thorough understanding of the expression of the antigens or their epitopes. Key early antigen targets were missing from the assays, contributing to insensitivity of serology in early infection. Also, cross-reactive epitopes are found in virtually all of the antigens used in the currently approved seroassays.
Read Also: What Medication Is Used For Lyme Disease
How To Test For Lyme Disease
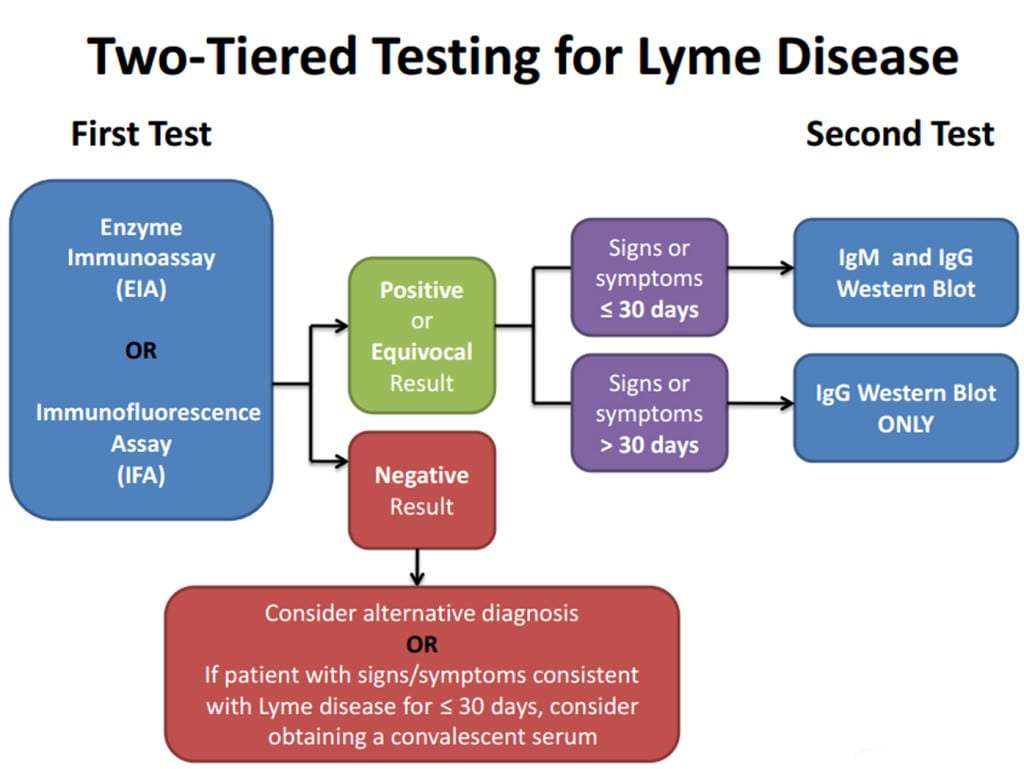
Wondering how to get tested for Lyme disease? The Centers for Disease Control and Prevention suggests a two-step testing process to check for a Lyme disease infection, both of which involve antibody testing , typically done on the same blood sample.
If the first step in the process returns a negative test result, then the second step is not necessary. However, if the first step yields a positive result, the second test is recommended as confirmation of a Lyme disease diagnosis. The Everlywell Lyme Disease Test follows this recommended protocol from the CDC, so it includes the two-step testing process.
Lyme Disease Laboratory Tests Available At Ripl
The primary service provided by RIPL is serological testing using well-characterised and validated screening and confirmatory tests in accordance with the NICE Lyme disease guideline.
RIPL participates in regular external quality assurance exercises as an independent measure of its performance.
Details of prices and turnaround times for Lyme testing are provided in Appendix 1 of the RIPL user manual.
Read Also: Do I Have Ms Or Lyme Disease
The Role Of Lyme Disease Tests
The purpose of the most common type of Lyme disease testing is to determine whether you have developed antibodies as a result of past exposure to the Borrelia bacteria that cause Lyme disease. Antibodies are proteins created by the immune system that target specific threats like bacteria and viruses.
Blood testing alone cannot determine whether you have Lyme disease. Instead, testing can provide helpful information that your doctor can consider along with other factors, such as any symptoms youve had and whether youve been exposed to ticks that can carry Borrelia, to determine if a diagnosis of Lyme disease is appropriate.
Beyond blood testing, it is possible to analyze fluid from the central nervous system for signs of the Borrelia bacteria.
Navigating Testing In Suspected Lyme Disease And The Approach I Use For Most Patients
Testing is a changing field in working up the possibility of symptoms being related to current or previous lyme infection. With the advent of increased awareness about the condition in general and recently the more readily availability of different testing methodologies from a variety of labs, it can be a confusion picture when information is sought beyond the standard two-tiered testing in acute lyme.
For many patients there is a basic relatively cost-effective approach that can be utilized. Testing is a complex process that can help to guide to find the most appropriate therapy. Testing is selected based on the history, exam and presentation of the patient. From a naturopathic perspective, it is just as important to evaluate the terrain of the patient in order to restore balance to physiology. The foundations of health need to be present, including most commonly optimal anti-inflammatory diet as well as sleep, support for weakened systems including assessment and removal of toxins . Testing for exposure to and active infection is one component of the process.
Also Check: Best Lyme Disease Doctors In Massachusetts
Current Approach To Laboratory Testing For Lyme Disease
Serologic assays are the most frequently used and familiar tests for the laboratory diagnosis of Lyme disease. At present in the United States, all tests currently cleared for diagnostic use by the US Federal Drug Administration are serologic assays. The current guidelines for serologic testing were adopted in 1994 . The challenge to the 1994 Dearborn Conference participants was to develop testing and interpretive guidelines that would standardize serologic testing for diagnostic purposes. The guidelines are geared to assess exposure to B. burgdorferi through the patients antibody response to infection rather than direct detection of nucleic acid or protein from the microbe. The limitations of serologic testing and advances were recently reported in detail .
Access To Lyme Disease Testing Services
This guidance on the laboratory diagnosis of Lyme disease is intended for healthcare professionals in the UK. Patients concerned about possible Lyme infection should consult an appropriate healthcare professional, for example their GP, in the first instance.
Health professionals wishing to discuss a possible case or ascertain local arrangements for testing should contact a local Infection specialist .
NHS testing for Lyme disease is available through local service providers and the Rare and Imported Pathogens Laboratory at PHE Porton where ISO15189 accredited confirmatory testing is also provided. RIPL is also introducing a testing service for neurological Lyme disease.
RIPL provides medical and laboratory specialist services to the NHS and other healthcare providers, covering advice and diagnosis of a wide range of unusual bacterial and viral infections, including Lyme disease.
RIPL continuously updates its methods and will make further information on Lyme disease diagnostic testing available as it arises.
Don’t Miss: Ozone Therapy For Chronic Lyme
Lyme Disease Test Two
Two-tiered Lyme disease testing uses two tests. The first is a screening test that should detect anyone who might have the disease. Tests that do this well have are regarded as having high sensitivity. This test is followed by a second test that is intended to make sure that only people with the disease are diagnosed. Tests that do this well have high specificity.
HIV/AIDS is diagnosed with tests that are both highly sensitive and highly specific. They are accurate more than 99% of the time. In Lyme disease, the second test is highly specific. So there are few false positives. Unfortunately, the screening test is highly insensitive and fails to accurately identify patients who have Lyme disease. The two-tiered test system misses roughly 54% of patients.
Because of this, LDo recommends the patients and physicians skip the ELISA and go straight to the Western blot.
Getting Tested For Lyme Disease
Lyme disease testing is usually ordered by a doctor and is used when there are signs or symptoms consistent with Lyme disease.
A blood sample can be drawn in a doctors office or other medical setting. If a test of cerebrospinal fluid is needed, an outpatient procedure called a lumbar puncture can be done in a hospital. Samples are then analyzed in a credentialed laboratory.
Recommended Reading: How Do They Test For Lyme Disease
Laboratory Tests That Are Not Recommended
Some laboratories offer Lyme disease testing using assays whose accuracy and clinical usefulness have not been adequately established. Examples of unvalidated tests include:
- Capture assays for antigens in urine
- Culture, immunofluorescence staining, or cell sorting of cell wall-deficient or cystic forms of B. burgdorferi
- Lymphocyte transformation tests
- In-house criteria for interpretation of immunoblots
- Measurements of antibodies in joint fluid
- IgM or IgG tests without a previous ELISA/EIA/IFA
How Will Pregnancy Affect Treatment For Lyme Disease

Early treatment of Lyme disease during pregnancy is important. Thats because if Lyme disease is left untreated, it can affect the placenta. According to the CDC , transmission of Lyme disease from mother to fetus is possible, though rare.
For pregnant people, treatment includes a round of antibiotics. Certain treatments for Lyme disease may not be used, as they can affect the fetus. If you suspect that you have Lyme disease, talk with a healthcare professional immediately.
Recommended Reading: Early Symptoms Of Lyme Disease
Types Of Lyme Disease Tests
Antibody titer tests for Lyme disease measure antibodies that are specific to the bacteria that can trigger the condition. Antibodies are part of the bodys immune response. Tests look for two types of antibodies, known as immunoglobulin M and immunoglobulin G .
IgM antibodies usually peak within a few weeks after an infection with Borrelia bacteria and start to collapse 4-6 months after infection. IgG antibodies are slower to develop, beginning to show 4-6 weeks after exposure and may peak 4-6 months after exposure. While IgM tends to be associated with an active infection, both IgM and IgG can persist in the blood for many years.
The Centers for Disease Control and Prevention recommends a two-stage or two-tiered test for these antibodies. If the initial blood test is positive for IgM or IgG antibodies associated with the Borrelia bacteria, a second test is done, often using different laboratory methods.
Testing for IgM and IgG can support the finding that you have been exposed to Borrelia, but this on its own does not prove whether or not you have Lyme disease.
Follow-up testing may be necessary if testing is inconclusive. This could involve repeat blood tests or the use of different types of medical tests to rule out other health conditions. In people suspected of bacterial reinfection, imaging tests may be used to detect joint inflammation.